Introduction
In January 2025, the FDA approved suzetrigine, a new non-opioid painkiller. It's cheap to manufacture, but like all new, on-patent drugs, it's expensive. Since expensive new drugs are restricted by Medicare, Medicaid, and insurance companies, suzetrigine (aka Journavx) won't become affordable or accessible for most patients until the patent expires in 2040. As a result, tens of millions of Americans will continue to receive unnecessary opioid pain prescriptions, and many of them will go on to develop an opioid addiction.
Instead of allowing this tragedy to needlessly grind forward, we should make suzetrigine low cost and first-line, through a public-private deal that would save payers money, ensure equal or better profits for the drugmaker Vertex, and reduce the number of opioid painkiller prescriptions written every year by up to 50%. This could be accomplished quickly.
Many, and Maybe Most, Opioid Addictions Begin with Prescriptions
Doctors and dentists in America prescribe about ~125M opioid pain prescriptions annually. While only a small percentage of these prescriptions lead to opioid addictions, that small percentage adds up to millions of people.
For any individual who develops an addiction after receiving a prescription, it's impossible to know whether they would have eventually become addicted anyway. But we have good reasons to believe that prescribing is a primary cause for many. And not only do patients themselves become addicted, but 60% of post-surgical opioid tablets go unused and sit in a medicine cabinet, often becoming a source of first exposure for others.
For a sense of scale:
- A 2025 survey of 1,515 US adults who reported non-prescription opioid use, predominantly illicit fentanyl, found 75% had started with prescription opioids:
- 39% first used opioids prescribed to them.
- 36% first used opioids prescribed to someone else.
- 25% first used illicit opioids, like heroin or non-prescribed fentanyl.
- ~80% of people with OUD in 2013 reported that their addiction started with prescription painkillers.
- These percentages have been fairly consistent since the 2000s.
- In 2023, 8.6 million Americans reported misusing prescription opioids.
- "Among US adults with any prescription opioid use in the past year, 7.0% met DSM‑5 criteria for prescription opioid use disorder" — Journal of Clinical Psychiatry 2024
Do We Need to Prescribe Pain Medicine at All?
Serious pain presents doctors and policymakers with a heart-wrenching dilemma. Pain can be life destroying. Should we err on the side of prescribing more opioids and accept addiction as a side effect? Or should we clamp down on prescribing and let more people live in pain? With suzetrigine reaching the market, the dilemma is closer to being resolved. There are several other non-addictive painkillers in pharma pipelines, so suzetrigine is likely the first of what will be a new generation of truly non-addictive pain medicines.
For milder and chronic pain, some experts, like Dr. Amy Baxter, make a compelling case that a) most post-operative pain passes quickly and does not need a painkiller prescription, b) medicalizing pain causes people to feel more pain, and c) opioids are not very effective for chronic pain or mild pain (see Dr. Baxter's TED Talk here). On the science, she's right. And yet, we still live in a society where most doctors want to give patients something after a procedure. There is limited top-down control over prescribing (for example, American dentists prescribe opioids ~30X more often than British dentists).
Could American doctors go cold turkey and simply stop prescribing pills for mild or transient pain? Perhaps. Some doctors are doing this by recommending acetaminophen and ibuprofen, which are often equally or more effective than hydrocodone. But many, many doctors still prescribe opioids. Whether it is objectively "necessary" to write these pain prescriptions, we are much more likely to rapidly reduce opioid prescribing if doctors and patients have a non-addictive alternative.
Decent Is Excellent
The approval of suzetrigine is not a normal situation.
Typically, a new medication is better than existing options for the use it is prescribed for, in this case pain reduction. But suzetrigine is actually not better than existing options at pain reduction. Its magic lies in what it lacks: addictiveness. Our medical insurance systems are built to balance the efficacy and cost of new treatments. They simply aren't structured to drive a rapid switch to a drug with higher cost and equivalent efficacy. That's why this case calls for an unusual intervention.
Suzetrigine, which acts on sodium channel pain-signaling, is not the strongest painkiller in the world, but it's about as strong as hydrocodone+acetaminophen (aka HB/APAP, aka Vicodin). It could replace many of, and perhaps even the majority, of opioid prescriptions. When a doctor or dentist feels compelled to give a patient something, suzetrigine can be that something.
Figure 1
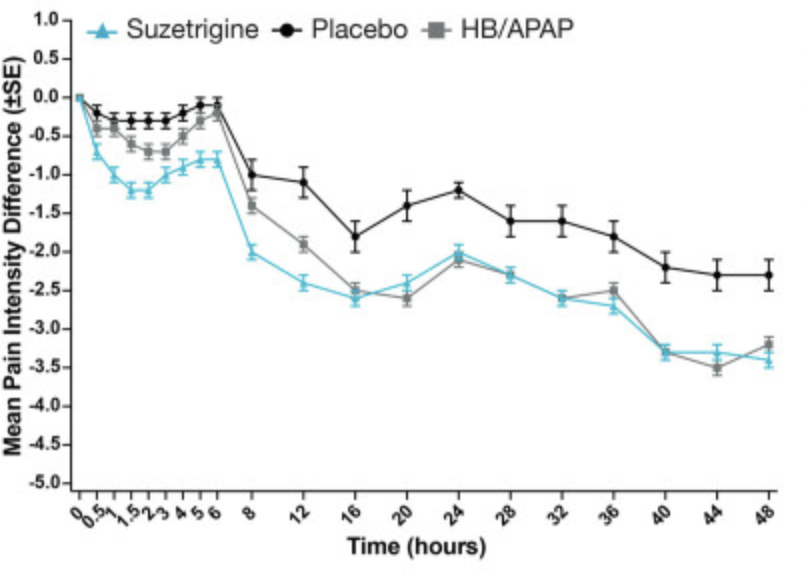
Suzetrigine alone shows pain reduction equivalent to HB+APAP
Source: Vertex Phase 3 Trial Data
As you can see above, while this is not a leap forward in pain reduction vs opioids, it is still a huge step for patient health: decent pain reduction with no addictive potential. In addition, the comparison here of suzetrigine vs hydrocodone+acetaminophen is a bit unfair: suzetrigine is also medically compatible with acetaminophen (the drugs work on different pathways) but was compared without acetaminophen. It is very likely that suzetrigine+acetaminophen would be even more effective than suzetrigine alone given its success in multimodal pain protocols. It's an easy combination to try.
How Many Opioid Prescriptions Could Be Avoided by Switching to Suzetrigine?
Roughly 50% of current opioid prescriptions could be replaced by suzetrigine based on our modeling. The use cases for suzetrigine are currently acute-pain only (a chronic pain label has not yet been approved) and fall on the milder side of the pain scale, which is where replacement is most likely.
Figure 2
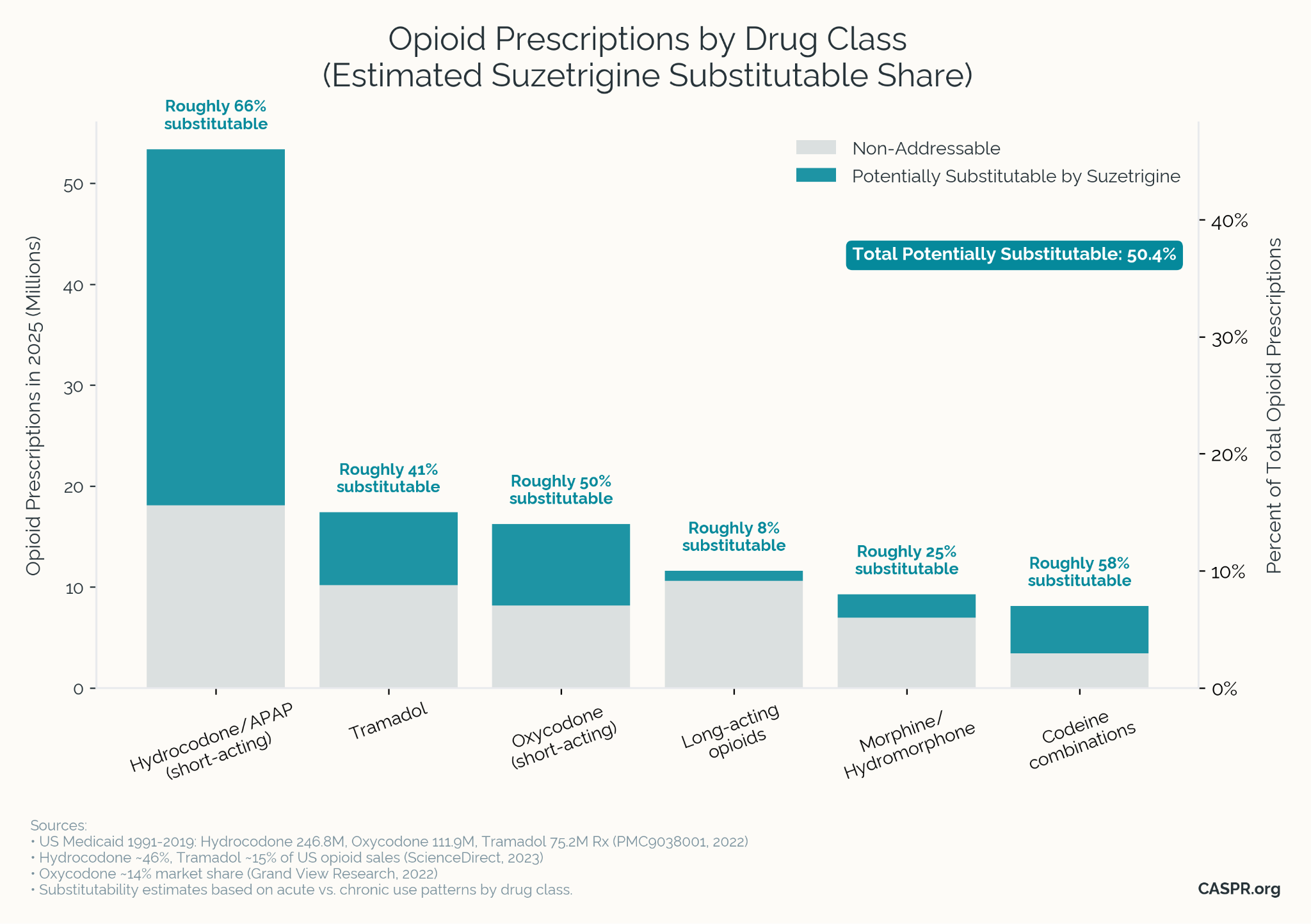
Hydrocodone + APAP (acetaminophen): ~68% substitutable (32M prescriptions per year)
- Primarily prescribed for short-term pain, which is suzetrigine's FDA-approved indication.
- Phase 3 trials show equivalent efficacy to hydrocodone/acetaminophen.
- Suzetrigine can be combined with acetaminophen for potentially even higher efficacy.
Tramadol: ~55% substitutable (15M prescriptions per year)
- Tramadol is a weaker opioid often used for moderate pain where suzetrigine may suffice.
- Similar indication profile as suzetrigine (acute musculoskeletal, post-procedural pain).
- Not yet fully substitutable because a) suzetrigine is not yet approved for chronic pain and b) tramadol's mechanism may work especially well for some patients.
Oxycodone (IR): ~45% substitutable (9M prescriptions per year)
- Immediate-release formulations are typically used for acute pain episodes.
- Post-surgical use overlaps with suzetrigine's approval and phase 3 trials.
- However, oxycodone is clearly stronger than suzetrigine and is used in chronic pain.
Codeine Combinations: ~60% substitutable (3M prescriptions per year)
- Frequently prescribed for mild-moderate pain and dental procedures.
- Short duration use aligns with suzetrigine's acute pain indication.
- Not a total substitute, because many codeine prescriptions are for cough suppression where suzetrigine is not likely to be effective.
These estimates are all based on the mix of acute vs chronic use of the milder opioid painkillers, given that suzetrigine is currently only approved for acute use. However, phase 3 trials of suzetrigine for chronic pain are currently in progress, and have a good chance at success, which would result in an approval in late 2026 or early 2027, greatly expanding the potential substitutability for opioids.
Current State of Suzetrigine Access
Suzetrigine is an expensive on-patent drug. Payers have erected a number of barriers to discourage use of suzetrigine and encourage doctors to try opioids first. Coverage is inconsistent and often complex for prescribers.
Most commercial plans place suzetrigine on Tier 3, the highest-cost tier, meaning high copays or deductibles if patients can even manage to get past prescribing restrictions. In Medicaid, only 19 states have removed prior authorization and step-edits as of mid-October 2025. Copays range from nominal amounts up to 20% of the state's cost (which can mean $50 for patients vs $5 for opioids). Medicare coverage is still being rolled out. VA access remains limited through clinical use rules.
Through year-end 2025, Vertex reported more than 550,000 suzetrigine prescriptions written and filled, a still-negligible fraction of the roughly 125M annual opioid prescriptions in the US. Vertex's January 2026 and February 2026 updates guided to more than triple that prescription volume in 2026. Even so, only a small percentage of patients today (~2-3%) are receiving suzetrigine instead of opioids, and we are needlessly continuing this prescription-to-addiction pipeline.
Making Suzetrigine First-Line Would Prevent Roughly ~100,000-160,000 New Opioid Addictions Per Year
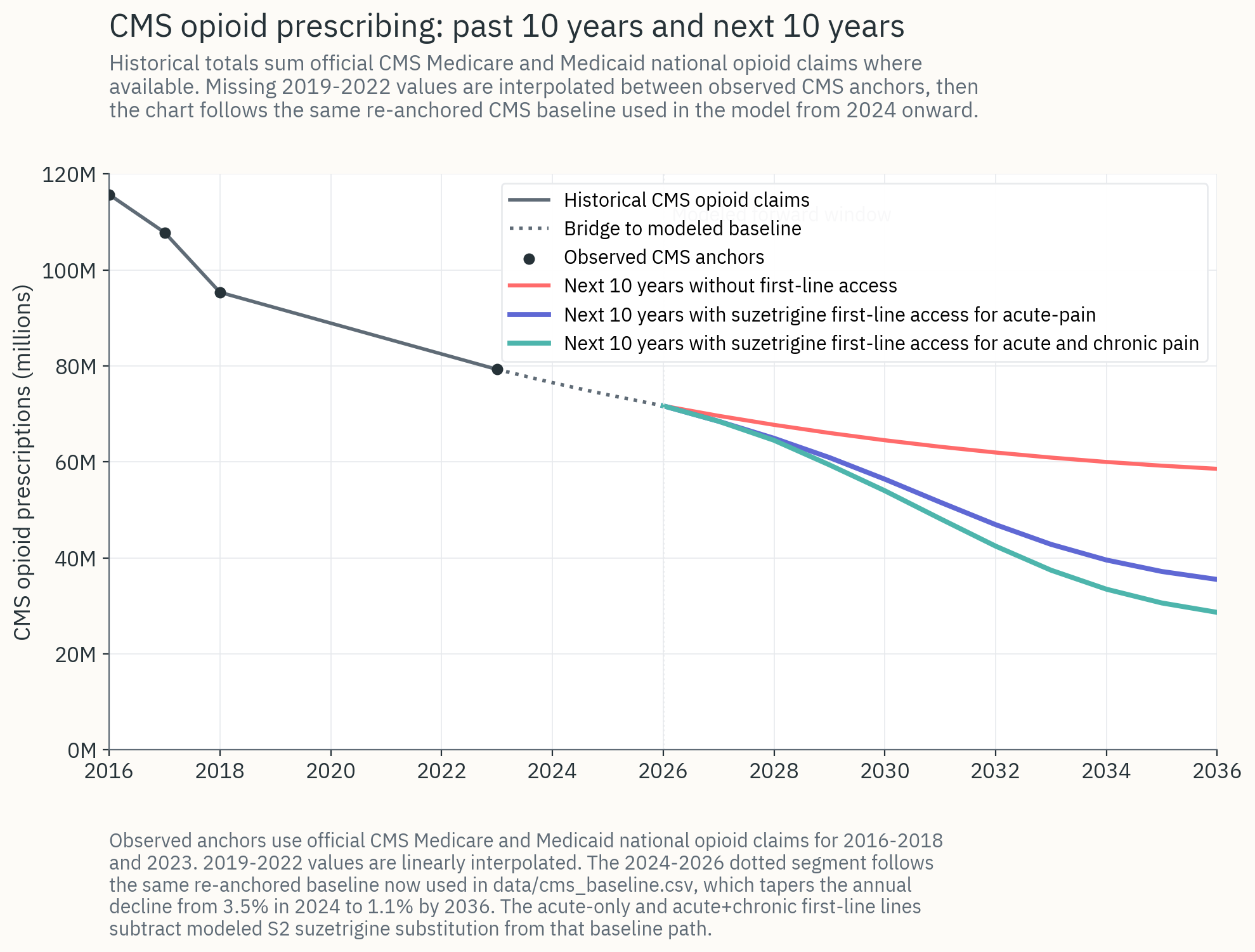
By agreeing to offer suzetrigine at low or no-cost to all applicable patients, the federal government could negotiate a dramatically lower price per pill with Vertex. This would cost public and private insurance companies roughly the same as what they would already pay for suzetrigine under the status quo (few suzetrigine patients, but high cost per patient) while replacing as much as 50% of all opioid prescriptions. In CASPR's fiscal modeling, making this switch saves CMS roughly $14.5 billion over 10 years in the primary $2/pill scenario, with most of the fiscal benefit coming from avoided OUD treatment costs.
The fiscal model uses a Bass-diffusion adoption curve calibrated to first-line access: under negotiated access, suzetrigine penetration of addressable CMS opioid prescriptions climbs toward roughly 85% by year 10, versus roughly 25% under managed-access status quo. That differential is what produces the incremental OUD prevention and fiscal savings reported here.
For a very rough ballpark, using the numbers we've looked at above:
- If ~75% of opioid addictions begin with prescription opioids, and
- If ~50% of opioid prescriptions can be replaced with suzetrigine prescriptions, mostly in acute pain today, then
- First-line acute-pain use could prevent ~60,000-80,000 new opioid addictions each year.
- If a chronic pain label is approved, chronic-pain use could prevent roughly another ~40,000-80,000 new opioid addictions each year.
- Combined, suzetrigine could prevent roughly ~100,000-160,000 of the ~435,000 new OUD cases in the US each year.
- Over a decade, this could be roughly ~1.0-1.6 million addictions avoided. Since opioid use and drug supply have significant network effects, the impact could be even larger, as prevalence becomes less common.
- The financial benefits would be tremendous. Roughly 5.7 million Americans have OUD. The economic and medical costs of the opioid epidemic are estimated to be well over $1 trillion per year. And the damage to families and communities goes far beyond what can be captured in economic numbers.
Tremendous Cost Savings in CMS
The downstream costs of OUD that are borne by Medicaid and Medicare make reductions in incidence highly cost effective. In CASPR's modeling, negotiating first-line access for suzetrigine at $2/pill yields large cost savings:
- Roughly +$14.5 billion in 10-year CMS net savings under the acute-pain label, the primary case modeled.
- Most of the savings come from avoided OUD care, with avoided direct opioid drug spend a smaller second-order contribution.
- If suzetrigine receives a chronic-pain label in 2027-28, the benefits roughly double to approximately +$29 billion in 10-year CMS net savings, because chronic-pain prescribing carries substantially higher per-course OUD incidence.
Acute vs. Chronic Use of Suzetrigine
Acute-pain label (currently approved): ~$14.5B net CMS savings over 10 years.
Chronic-pain label (expected 2027-2028): roughly ~$29B net CMS savings, with OUD prevention benefits approximately doubling.
Figure 3
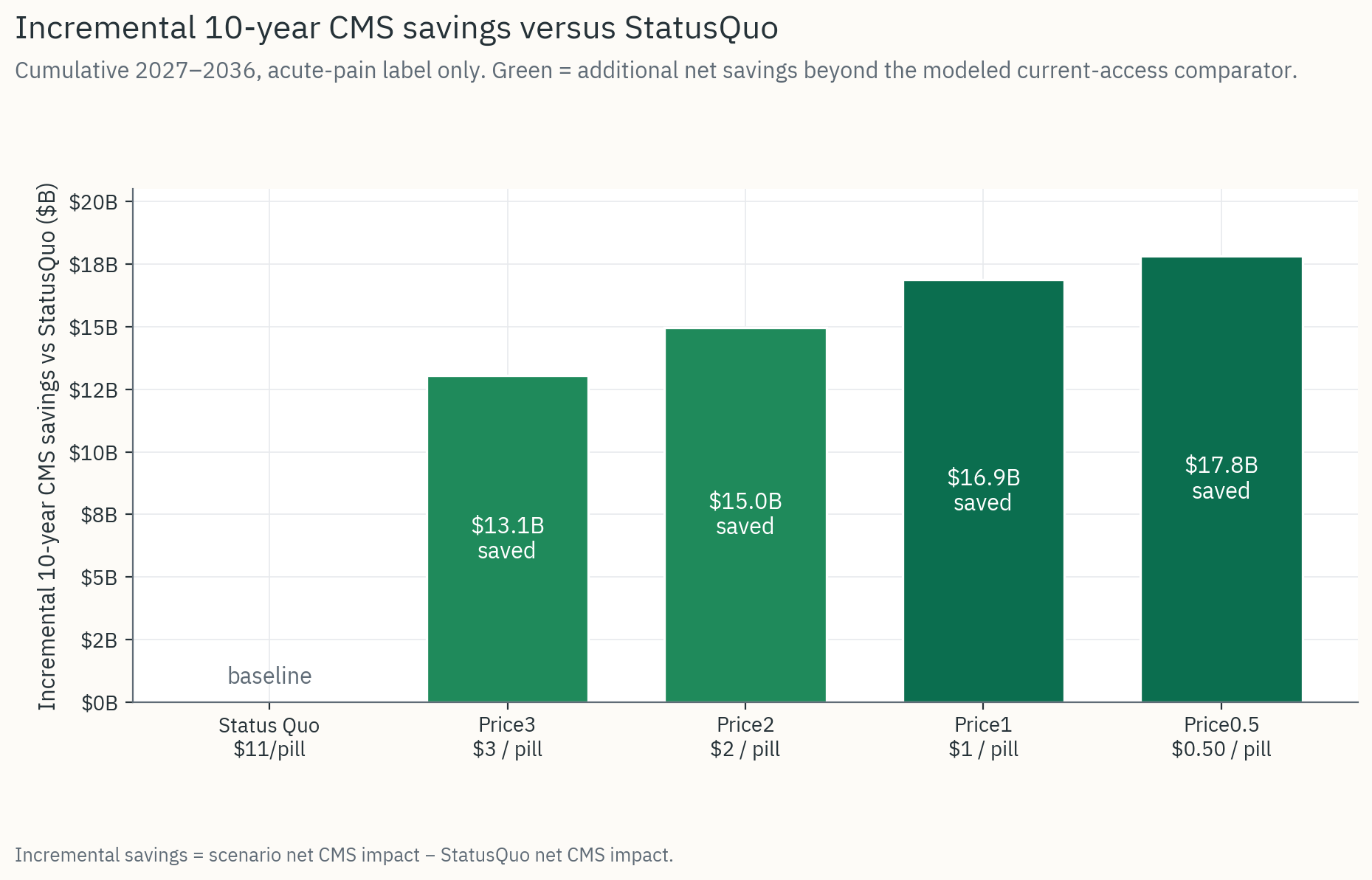
Incremental 10-year CMS savings versus status quo, acute-pain label only, 2027-2036.
Source: CASPR Fiscal Analysis
Making a Deal
The current Administration has shown openness to deal-making that reduces costs or improves access to medicine, like the recent GLP‑1 pricing agreements with Novo Nordisk and Eli Lilly. The case for a suzetrigine deal is even stronger. By agreeing to offer suzetrigine at low or no cost to all applicable patients in government health systems, and with a structure to enable private insurance companies to do the same, the government could negotiate a far lower price per pill. It could rapidly bring an effective non-opioid option for acute pain to far more Americans, while holding patient out-of-pocket costs at zero and giving Vertex revenue equal or greater to their earnings without such an arrangement. Combined with a provider-awareness push, opioid prescribing could be dramatically reduced in a matter of months.
This deal would also serve as a template for the future, as additional non-addictive painkillers come to market, ensuring that the pharma development pipeline remains strong and pricing is competitive and fair for the public.
An agreement along these lines would cover:
- Federal delivery systems (VA, DoD, and IHS). The VA has already cut opioid prescribing by roughly 67% since 2012, which caps incremental volume there, but IHS serves American Indian and Alaska Native populations with some of the highest opioid-attributable harm rates in the country, and DoD carries a younger, surgically intensive population where acute-pain substitution has direct value.
- Medicare fee-for-service and participating Medicare Advantage plans through a CMS Innovation Center model.
- Highly appealing for states to opt-in via a standardized Medicaid template.
- A buy-in option for private insurance to adopt a similar deal, with incentives to do so. Commercial adoption matters even where per-member OUD-prevention value is diluted by churn, because commercial prescribing behavior tracks CMS formulary norms over time.
The fiscal-analysis companion includes a detailed appendix on federal-channel and commercial spillover dynamics, and on how these channels compound the acute-label CMS savings shown here.
For each of these systems, there are structural nuances and details to negotiate, but it would all flow naturally as long as it focuses on these core objectives:
- Broad first-line access for suzetrigine
- Make it as easy or easier to prescribe than opioids.
- Make it easy to access (no prior auths, no step therapy).
- Make the co-pays as cheap or cheaper than opioids.
- Low price
- In exchange for this massive increase in patient access, Vertex prices suzetrigine competitively with opioids (70-80% reduction from list price).
A Victory for Americans and for Vertex
Summarizing the impact and opportunity:
- Currently, suzetrigine lists for $15.50 per pill vs ~$0.35 for generic hydrocodone+APAP. Therefore insurers put up barriers to reduce usage.
- Through a deal to make suzetrigine dramatically cheaper and easily available to everyone, Vertex can earn comparable or greater revenue over the patent window by reaching many more patients sooner.
- Millions of opioid addictions will be prevented. This will save federal, state, and local governments billions a year in direct and indirect savings.
- In CASPR's modeling, Medicare and Medicaid-linked systems save roughly +$14.5 billion over 10 years in the core negotiated acute-pain case, with chronic-label expansion roughly doubling the fiscal savings.
- The total economic burden of opioid addiction and overdose has been estimated at roughly $1.5-2.7 trillion annually for recent years (2017 and a rough extrapolation to 2023). Even a modest reduction in OUD prevalence would drive hundreds of billions in fiscal savings and reduced economic burden.
- The personal and emotional benefits for families and the social benefits to communities would be immeasurable.
Under this structure, Vertex doesn't get a handout but does get a predictable revenue stream that is at least as attractive as the typical high-price but low volume pathway they are currently on. In the process, Vertex also gains a national showcase role as the company that led a transformation in rates of addiction.
The Administration, in turn, secures a groundbreaking deal that quickly and measurably drives down opioid use and new opioid addictions without raising costs and while increasing patient choice. This model could then be extended to other drug companies as additional new non-opioid painkillers come to market.
Further Reading
A 10-year CMS fiscal model of a negotiated suzetrigine access deal. Includes five pricing scenarios, Bass-diffusion adoption curves, OUD cases and OUD-attributable deaths prevented, federal-channel appendix (VA, DoD, IHS), commercial spillover, Vertex revenue view, and full assumption tables.